
If the central atom does not have any lone pairs (or a single electron), the molecular geometry is the same as the electron pair geometry. The molecular geometry only takes into consideration the number of bonds. The electron pair geometry is determined by the total number of charge clouds around an atom. Looking at the water molecule, we see the oxygen has 4 electron groups (charge clouds) surrounding it, two H-O bonds and two lone pairs of electrons. These electron groups are referred to as charge clouds. A single bond, double bond, triple bond, a lone pair, or a single electron count as a single electron group. 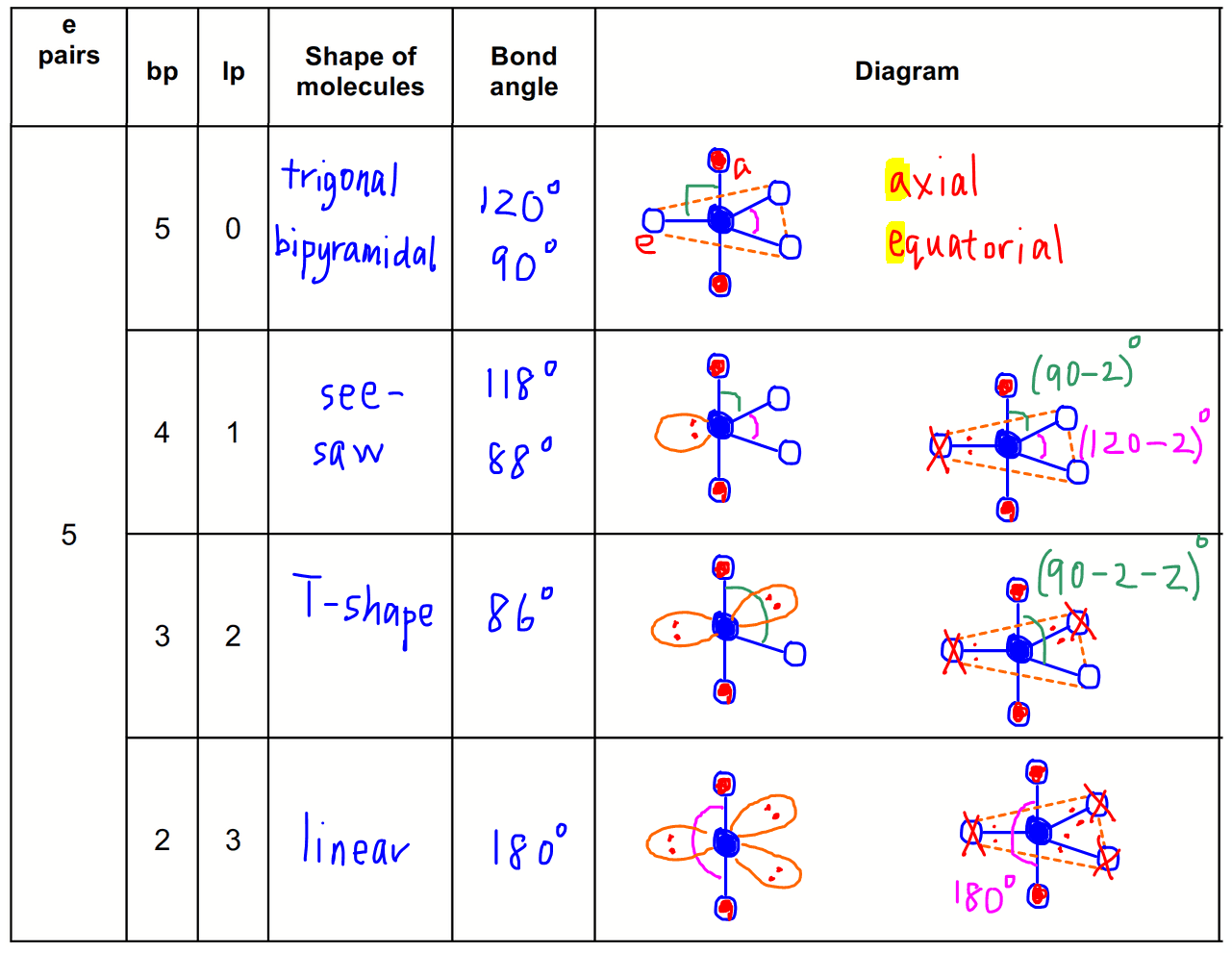
Here we will discuss both electron pair geometries (also called electron group or electron domain geometries) and molecular geometries (also called molecular shapes).Ī group of electrons is any number of electrons that occupies a localized region around an atom. We use the Valence Shell Electron Pair Repulsion (VSEPR) model to predict the shapes of molecules. The regions of electrons density, bonds and nonbonding electrons, will repel one another and therefore orient as far away from each other as they possibly can to minimize these repulsions. If four balloons are tied together, each balloon will orient to a corner of a tetrahedron with angles of 109.5 o.Įlectrons in molecules will act similarly to the balloons. If we add a third balloon, we see the balloons point to the corners of an equilateral triangle with angles of 120 o apart. In fact, the balloons are 180 o apart and have a linear orientation. The balloons will point away from one another as shown in the figure below. Imagine two equal sized balloons that are tied together. Molecules and ions are three dimensional and we have only learned how to draw two dimensional shapes. Lewis structures do not tell us anything about molecular shape. Lewis structures were used previously to determine how the atoms are connected in a molecule and show where the lone pairs of electrons are located.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
